 These three electronic states differ only in the spin and the occupancy of oxygen's two antibonding g -orbitals, which are degenerate (equal in energy).  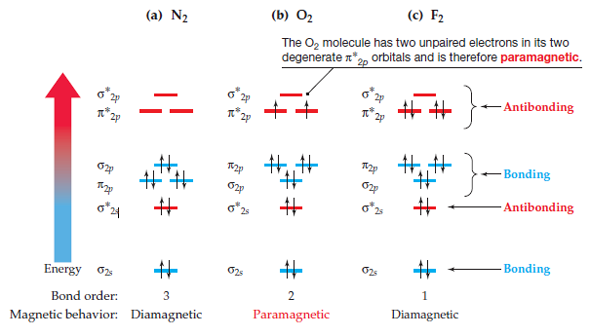
Note: -Electronic configuration of molecular orbital must be according to Hund’s maximum multiplicity rule, according to which the orbital available in the subshell of a molecule are first filled singly with parallel spin electron before they begin to pair and subshell give maximum number of unpaired electron with parallel spin.\( \newcommand\): Hand-drawn molecular orbital diagram for dioxygen. Molecular orbital theory predicts the electronic ground state denoted by the molecular term symbol 3 g, and two low-lying excited singlet states with term symbols 1 g and 1 + g. Energy level diagram for Molecular orbitals 1) Stability of molecules in terms of bonding and antibonding electrons 2) Stability of molecules in terms of bond. Atomic valence electrons (shown in boxes on the left and right) fill the lower-energy molecular orbitals before the higher ones, just as is the case for atomic orbitals. 9.10: Molecular Orbital Theory Predicts that Molecular Oxygen is. Answer Verified 249. Compare the bond order to that seen in the Lewis structure. Draw a molecular orbital diagram of N2 N 2 or O2 O 2 with magnetic behavior and bond order. It is recommended to name the SVG file Oxygen molecule orbitals diagram.svg then the template Vector version available (or Vva) does not need the new. HF nb Energy H 13.6 eV 1s F 18.6 eV 40. We represent this configuration by a molecular orbital energy diagram ( link ) in. Construct a qualitative molecular orbital diagram for peroxide anion, O 2 2. This theory explained the paramagnetic nature of $\text$. Molecular Orbital Diagrams This scheme of bonding and antibonding orbitals is usually depicted by a molecular orbital diagram such as the one shown here for the dihydrogen ion H 2 +. MO Diagram for HF The AO energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. (2s)2( 2s)2(2pz)2(2px)2(2py)2( 2px)1 Your first answer option is for N 2. The electron would be removed from the pi orbital, as this is the highest in energy. 
Hint: Molecular orbital theory was put forward by Hund and Mulliken, which can be applied to explain the properties, that was not explained by Valence bond theory. Paramagnetic We can work this out by looking at the molecular orbital diagram of O2 O2+ has 1 fewer electron than O2 which is what gives it the positive charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
